5828. Melphalan
Nomenclature
Description and references
Bifunctional, non-cell-cycle specific alkylating agent. Syntheses: Bergel, Stock, J. Chem. Soc. 1954, 2409; 1955, 1223; eidem, US 3032584; US 3032585 (both 1962 to NRDC); Larionov, Lancet 2, 169 (1955). Neurotoxicity study: M. G. Donelli et al., J. Pharm. Pharmacol. 18, 760 (1966). HPLC determn in biological fluids: I. Muckenschnabel et al., Eur. J. Pharm. Sci. 5, 129 (1997). Review of carcinogenicity studies: IARC Monographs 9, 167-180 (1975). Review: R. L. Furner, R. K. Brown, Cancer Treat. Rep. 64, 559-574 (1980). Clinical trial in multiple myeloma: A. Palumbo et al., Blood 104, 3052 (2004). Review of pharmacology and clinical efficacy: B. L. Samuels, J. D. Bitran, J. Clin. Oncol. 13, 1786-1799 (1995).
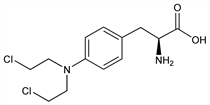
Properties
Needles from methanol (monosolvate), mp 182-183° (dec). [α]D25 +7.5° (c = 1.33 in 1.0N HCl); [α]D22 31.5° (c = 0.67 in methanol). Soluble in ethanol, propylene glycol. Practically insol in water, chloroform, ether. LD50 i.p. in rats: 14.7 μmol/kg. See: W. C. J. Ross, Biochem. Pharmacol. 13, 969 (1964).Derivative
d-Form.Nomenclature
Properties
Needles from methanol (monosolvate), mp 181.5-182° (dec). [α]D21 7.5° (c = 1.26 in 1.0N HCl).Derivative
dl-Form.Nomenclature
Properties
Tiny needles from methanol, mp 180-181°.Caution
This substance is listed as a known human carcinogen: Report on Carcinogens, Eleventh Edition (PB2005-104914, 2004) p III-164.Therapeutic Category
Antineoplastic.
Therapeutic Category (Veterinary)
Antineoplastic.Keywords
Antineoplastic; Alkylating Agents; Nitrogen Mustards