5837. Menthol
Nomenclature
CAS number: 2216-51-5;
89-78-1
(dl-Form)
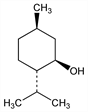
(1R,2S,5R)-5-Methyl-2-(1-methylethyl)cyclohexanol; 3-p-menthanol; l-menthol; hexahydrothymol; peppermint camphor. C10H20O; mol wt 156.27.
C 76.86%, H 12.90%, O 10.24%.
Description and references
Obtained from peppermint oil or other mint oils, or prepd synthetically by hydrogenation of thymol. Chromatographic sepn from mint oils: Chang, US 2760993 (1956 to Iowa State College). Toxicity data: P. M. Jenner et al., Food Cosmet. Toxicol. 2, 327 (1964) DOI.
Properties
Crystals or granules; peppermint taste and odor. d 0.890. mp 41-43°. bp 212°. nD25 1.458. [α]D18 50° (10% alc soln). Slightly sol in water; very sol in alcohol, chloroform, ether, petr ether; freely sol in glacial acetic acid, liquid petrolatum. LD50 orally in rats: 3180 mg/kg (Jenner). Incompat: Butylchloral hydrate, camphor, phenol, chloral hydrate, Exalgine, betanaphthol, resorcinol or thymol in triturations; potassium permanganate, chromium trioxide, pyrogallol.Use
In liqueurs, confectionery, perfumery, cigarettes, cough drops, and nasal inhalers.Therapeutic Category
Antipruritic (topical).
Therapeutic Category (Veterinary)
Has been used as a mild local anesthetic, antiseptic; internally as a carminative and gastric sedative.Keywords
Antipruritic