5846. Meparfynol
Nomenclature
Description and references
Prepd from methyl ethyl ketone and sodium acetylide in liquid ammonia or by a Grignard reaction: DE 285770 (1913 to Bayer); also DE 289800 and DE 291185 see Frdl. 12, 55, 56, 57; Sung Wouseng, Ann. Chim. [10] 1, 343 (1924); Rupe, Vonaesch, Ann. 442, 80 (1925); Carothers, Coffman, J. Am. Chem. Soc. 54, 4071 (1932); Campbell et al., ibid. 60, 2882 (1938); Campbell, Campbell, Proc. Indiana Acad. Sci. 50, 123-127 (1940), C.A. 35, 5471; Smith, US 2385547 (1945 to Commercial Solvents); A. W. Johnson, The Chemistry of the Acetylenic Compounds vol. I (Edward Arnold & Co., London, 1946) p 278; Hurd, McPhee, J. Am. Chem. Soc. 69, 239 (1947); P. Piganiol, Acetylene Homologs and Derivatives (Brooklyn, 1950); Papa et al., Arch. Biochem. Biophys. 33, 482 (1951); Thiele, Martinez, Ciencia (Mexico City) 15, 70 (1955). An optically active form has been described by Hickmann, Kenyon, J. Chem. Soc. 1955, 2051. Toxicity data: Margolin et al., Science 114, 384 (1951).
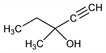
Properties
Mobile liquid. Acrid odor, burning taste. d420 0.8688. d2020 0.8721; 7.28 lbs/U.S. gal. bp760 121-122°; bp37 50°; bp6.5 20°. mp 30.6°. nD20 1.4318. Flash pt 101.3°F. Surface tension at 25°: 23.8 dynes/cm; 5% aq soln: 34.1 dynes/cm. Soly in water at 25°: 12.8 g/100 ml. Sol in ether. Miscible with acetone, benzene, carbon tetrachloride, Cellosolve, cyclohexanone, diethylene glycol, ethyl acetate, kerosene, methyl ethyl ketone, mineral spirits, ethanolamine, neatsfoot oil, petr ether, soybean oil, Stoddard solvent. LD50 orally in mice, rats, guinea pigs: 600-900 mg/kg (Margolin).Derivative
Carbamate.Nomenclature
Description and references
Prepn: McLamore et al., J. Org. Chem. 20, 1379 (1955); from 3-methyl-1-pentyn-3-ol, trichloroacetic acid, KOCN: McCrea et al., GB 761817 (1956 to British Schering). Acute toxicity: Soehring et al., Arzneim.-Forsch. 5, 161 (1955).
Properties
Crystals from ether + petr ether. mp 55.8-57° (McLamore); from cyclohexane, mp 53.5-55° (McCrea). bp16 120-121°; bp0.01 95°. Solubility in water: 1.6 g/100 ml. LD50 (4 hr) s.c. in mice: 0.56 g/kg (Soehring).Therapeutic Category
Hypnotic; sedative.
Keywords
Sedative/Hypnotic; Alcohols