5910. Mesoridazine
Nomenclature
Description and references
Dopamine receptor blocking agent; analog of thioridazine. Prepn: Renz et al., US 3084161 (1963 to Sandoz). Pharmacology and toxicology: Loew et al., Boll. Chim. Farm. 106, 332-371 (1967). Effects on dopaminergic function in comparison with sulforidazine and thioridazine, q.q.v.: D. M. Niedzwiecki et al., J. Pharmacol. Exp. Ther. 228, 636 (1984); C. D. Kilts et al., ibid. 231, 334 (1984). GLC determn in plasma: E. C. Dinovo et al., J. Pharm. Sci. 65, 667 (1976). Clinical evaluation in sleep disorders: K. Adam et al., Br. J. Clin. Pharmacol. 3, 157 (1976); as antipsychotic: R. Axelsson, Curr. Ther. Res. 21, 587 (1977). Toxicity studies: S. Maruyama et al., Niigata Igakkai Zasshi 81, 611 (1967), C.A. 68, 76856h (1968). Brief description: J. Am. Med. Assoc. 216, 313 (1971).
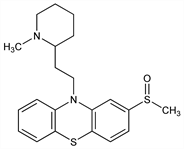
Properties
Oily product.Derivative
Benzenesulfonate.Nomenclature
Properties
LD50 in mice (mg/kg): 33 i.v.; 611 s.c.; 346 orally (Maruyama).Derivative
Tartrate.Properties
Crystals from ethyl acetate, mp 115-120°.Therapeutic Category
Antipsychotic.
Keywords
Antipsychotic; Phenothiazines