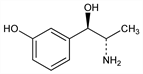
5932. Metaraminol
Nomenclature
CAS number: 54-49-9
(αR)-α-[(1S)-1-Aminoethyl]-3-hydroxybenzenemethanol; ()-α-(1-aminoethyl)-m-hydroxybenzyl alcohol; m-hydroxynorephedrine; m-hydroxypropadrine; m-hydroxyphenylpropanolamine; m-hydroxy-α-(1-aminoethyl)benzyl alcohol; 2-amino-1-(m-hydroxyphenyl)-1-propanol; 1-(m-hydroxyphenyl)-2-amino-1-propanol; α-(m-hydroxyphenyl)-β-aminopropanol; metaradrine. C9H13NO2; mol wt 167.21.
C 64.65%, H 7.84%, N 8.38%, O 19.14%.
Description and references
Sympathomimetic amine. Prepn from m-hydroxyisonitrosopropiophenone: GB 353361 (1930 to I. G. Farben); l-form: GB 396951 (1932 to I. G. Farben); from l-m-hydroxyphenylacetylcarbinol: CH 162367 (1931 to I. G. Farben); see also US 1948162 and US 1951302; Hartung, US 1995709 (1935 to Sharp & Dohme). HPLC determn in commercial formulations: C. J. Martin, S. J. Saxena, J. Pharm. Sci. 69, 1459 (1980). Pharmacology: A. Cession-Fossion, Arch. Int. Pharmacodyn. Ther. 172, 421 (1968). Toxicity data: O. H. Siegmund et al., J. Pharmacol. Exp. Ther. 92, 207 (1948). Mechanism of action: D. C. Harrison et al., Ann. Intern. Med. 59, 297 (1963).
Derivative
Bitartrate.Nomenclature
CAS number: 33402-03-8
Aramine (Merck & Co.); Araminon (Banyu). C13H19NO8; mol wt 317.29.
C 49.21%, H 6.04%, N 4.41%, O 40.34%.
Properties
White, crystalline powder, mp 176-177°. Freely sol in water; slightly sol in alcohol. Practically insol in chloroform and ether. pH of 1% aq soln about 3.5.Derivative
Hydrochloride.Nomenclature
CAS number: 5967-52-2
C9H13NO2.HCl; mol wt 203.67.
C 53.07%, H 6.93%, N 6.88%, O 15.71%, Cl 17.41%.
Properties
Hygroscopic crystals, [α]D20 19.75°. Freely sol in water. LD50 i.p. in mice: 440 mg/kg (Siegmund).Derivative
Oxalate dihydrate.Nomenclature
CAS number: 5967-53-3
C9H13NO2.C2H2O4.2H2O; mol wt 293.27.
C 45.05%, H 6.53%, N 4.78%, O 43.64%.
Properties
Crystals, mp 190°. [α]D20 21.66°. Sol in water.Therapeutic Category
Adrenergic.
Keywords
α-Adrenergic Agonist; Antihypotensive