5941. Methacrylic Acid
Nomenclature
CAS number: 79-41-4
2-Methyl-2-propenoic acid; α-methylacrylic acid. C4H6O2; mol wt 86.09.
C 55.81%, H 7.02%, O 37.17%.
Description and references
Occurs in oil from Roman chamomile. Prepd by dehydration of α-hydroxyisobutyric acid: Crawford, US 2143941 (1939 to I.C.I.); by hypochlorite oxidation of methyl α-alkylvinyl ketone: Meitzner, US 2192142 (1940 to Rohm & Haas); by hydrolysis of acetone cyanohydrin: Crawford, GB 405699 (1932 to I.C.I.); by oxidation of methacrolein: Bauer, US 2153406 (1939 to Rohm & Haas). Toxicity study: W. Deichmann, J. Ind. Hyg. Toxicol. 23, 343 (1941). Review: J. W. Nemec, L. S. Kirch in Kirk-Othmer Encyclopedia of Chemical Technology vol. 15 (Wiley-Interscience, New York, 3rd ed., 1981) pp 346-376.
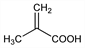
Properties
Long prisms, mp 16°. Acrid, repulsive odor. d420 1.0153. bp760 163°; bp30 81°; bp12 63°. nD20 1.43143. Flash pt, open cup: 170°F (76°C). Corrosive. Sol in warm water; miscible with alc, ether. Polymerizes easily, especially on heating or in the presence of traces of HCl. The polymer forms a ceramic-looking mass, sol in abs alc, from which it is precipitated by ether.Derivative
Methyl ester.Nomenclature
CAS number: 80-62-6
Methyl methacrylate. Description and references
Review of carcinogenic risk: IARC Monographs 60, 445-474 (1994).