5966. Methenamine
Nomenclature
Description and references
Prepn: A. Butlerow, Ann. 115, 322 (1860); from formaldehyde and ammonia: F. Meissner, E. Schwiedessen, US 2762799; US 2762800 (both 1956); Faith, Keyes & Clark's Industrial Chemicals, F. A. Lowenheim, M. K. Moran, Eds. (Wiley-Interscience, New York, 4th ed., 1975) pp 445-448. Crystal property studies: G. H. Heilmeier, Appl. Opt. 3, 1281 (1964). Comprehensive reviews: J. F. Walker, Formaldehyde (Reinhold, New York, 3rd ed., 1964) pp 511-551; J. M. Dreyfors et al., Am. Ind. Hyg. Assoc. J. 50, 579-585 (1989). Review of therapeutic use: R. Gleckman et al., Am. J. Hosp. Pharm. 36, 1509-1512 (1979); of toxicology: H. J. Trochimowicz et al. in Patty's Industrial Hygiene and Toxicology vol. 2E, G. D. Clayton, F. E. Clayton, Eds. (John Wiley & Sons, New York, 1994) pp 3324-3327.
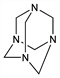
Properties
Colorless, odorless crystalline solid with sweet, metallic taste. Sublimes at about 263° without melting and with partial decompn; somewhat volatile at lower temp. nD25 1.5911. Flammable. Specific heat: 36.4 cal/°C. d 1.331. Soly at 20° (g/100g): chloroform 13.4; methanol 7.25; abs ethanol 2.89; acetone 0.65; benzene 0.23; xylene 0.14; ether 0.06. Practically insol in petr ether. pH of 0.2 molar aq sol 8.4.Derivative
Anhydromethylenecitrate.Nomenclature
Properties
White, cryst powder. mp 175° with decompn. Sol in 10 parts water; very slightly sol in alcohol, ether.Derivative
Hippurate.Nomenclature
Properties
Crystals, mp 105-110°. Freely sol in water, alcohol.Derivative
Mandelate.Nomenclature
Properties
Crystals or white, crystalline powder. mp 128-130°. Slightly sol in acetone, ether; sol in alcohol, chloroform; very sol in water.Derivative
Sulfosalicylate.Nomenclature
Properties
Occurs as the monohydrate. Odorless, cryst powder, mp ≈190° with decompn. Sol in 8 parts water; slightly sol in alcohol.Use
In adhesives, coatings, and sealing compounds; as cross-linking agent for hardening phenol-formaldehyde resin and vulcanizing rubber; as corrosion inhibitor for steel; as fuel tablets for camping stoves; as stabilizer for lubricating and insulating oils; for the manufacture of explosive compounds (see Cyclonite); antimicrobial food additive; detection of metals.Therapeutic Category
Antibacterial (urinary).
Therapeutic Category (Veterinary)
Antibacterial (urinary).Keywords
Antibacterial (Synthetic)