5992. Methoxyethylbenzeneboronic Acid
Nomenclature
CAS number: 159752-39-3
[2-[(1R)-1-Methoxyethyl]phenyl]boronic acid. C9H13BO3; mol wt 180.01.
C 60.05%, H 7.28%, B 6.01%, O 26.66%.
Description and references
Synthesis of (+)-form and use: K. Burgess, A. M. Porte, Angew. Chem. Int. Ed. 33, 1182 (1994). Chemoenzymatic stereosynthesis of both enantiomers and use: S. M. Resnick et al., J. Org. Chem. 60, 3546 (1995).
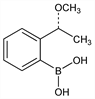
Properties
White solid from n-hexane, mp 72-74°. [Ga]D25 +27 (c = 0.5 in CH2Cl2) (Burgess); [α]D +27.2° (c = 4.6 in CH2Cl2) (Resnick).Derivative
()-S-Form.Nomenclature
CAS number: 166191-23-7