Nomenclature
CAS number: 96-33-3
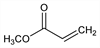
2-Propenoic acid methyl ester; acrylic acid methyl ester; methyl propenoate.
C
4H
6O
2; mol wt 86.09.
C 55.81%, H 7.02%, O 37.17%.
Description and references
Convenient prepn from ethylene chlorohydrin:
DE 571123 (1928 to Rohm & Haas). For direct syntheses from acetylene
and carbon monoxide see W. Reppe, Chemie & Technik der Acetylen-Druck-Reaktionen (Weinheim,
2nd ed., 1952). Toxicity study: H. F.
Smyth, C. P. Carpenter, J. Ind. Hyg. Toxicol. 30, 63 (1948).
Properties
Liquid. Acrid odor. Lacrimator. d420 0.9561; d2020 0.9574; d40 0.9702; d45 0.9868; d410 0.9929. Weighs
8.0 lbs/gal. mp 76.5°. bp608 70°; bp428 60°; bp298 50°; bp200 40°; bp88 20°; bp54 10°; bp41.5 5°; bp32 0°; bp24.5 5°; bp18.5 10°. nD20 1.401. Sp heat at 60° = 0.444 cal/g/°C;
heat of vaporization 8.25 kcal/mol; heat of combustion 502.88 kcal/mol.
Soly in water at 20° = 6 g/100 ml, at 40° = 5 g/100 ml. Soly of
water in methyl acrylate at 20° = 1.8 ml/100 g. Sol in alc, ether.
Azeotropes: 9.5% water = bp 73°; 49.0% methanol = bp 61°. Easily polymerizes on standing. The polymerization process can
be speeded up by heat, light, and peroxides. If pure, the monomer
can be stored below +10° without incurring polymerization. LD50 orally in rats:
0.3 g/kg (Smyth,
Carpenter).Derivative
Polymer.
Properties
Transparent, elastic substance. Practically no
odor. Little adhesive power. Resists the usual solvents.Caution
Potential symptoms of overexposure
to the monomer are irritation of eyes, upper respiratory system and
skin. See NIOSH Pocket Guide to
Chemical Hazards (DHHS/NIOSH 97-140, 1997) p 198. See also Clinical Toxicology of Commercial
Products, R. E. Gosselin et al., Eds. (Williams
& Wilkins, Baltimore, 5th ed., 1984) Section II, p 409.Use
The monomer in manuf leather finish resins, textile
and paper coatings, and plastic films. Produces the hardest resin
of the acrylate ester series.
