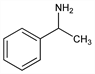
6026. α-Methylbenzylamine
Nomenclature
Description and references
Prepn from acetophenone and ammonium formate: A. W. Ingersoll et al., J. Am. Chem. Soc. 58, 1808 (1936); by reduction of acetophenone in liquid ammonia: J. C. Robinson, H. R. Snyder, Org. Synth. coll. vol. III, 717 (1955). Stereospecific synthesis of R-(+)-form: H. Takahashi et al., Chem. Pharm. Bull. 29, 3387 (1981). Resolution of (dl)-methylbenzylamine: W. Theikecker, H.-G. Winkler, Ber. 87, 690 (1954). Use of (R)-(+)-methylbenzylamine as resolving agent: E. J. Corey, J. Mann, J. Am. Chem. Soc. 95, 6832 (1973). Use as chiral intermediate in synthesis of α-methyl-α-amino nitriles: K. Weinges et al., Ber. 110, 2098 (1977); of β-amino acids: M. Furukawa et al., Chem. Pharm. Bull. 26, 260 (1978). Toxicity: H. F. Smyth et al., Arch. Ind. Hyg. Occup. Med. 4, 119 (1951).