Nomenclature
CAS number: 78-93-3
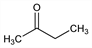
2-Butanone; ethyl methyl ketone; MEK; 2-oxobutane.
C
4H
8O; mol wt 72.11.
C 66.62%, H 11.18%, O 22.19%.
Description and references
Prepn from ethyl 2-methylacetoacetate: J.
Schramm, Ann. 398,
242 (1913). Manuf by dehydration of 2-butanol and by catalytic oxidation
of n-butenes: A. J. Papa, P. D. Sherman, Jr. in Kirk-Othmer Encyclopedia of Chemical Technology vol. 13 (Wiley-Interscience, New York, 3rd ed., 1981)
pp 903-907. Toxicity: H. F. Smyth et al., Am. Ind. Hyg. Assoc. J. 23, 95
(1962). Review of toxicology and human exposure: Toxicological Profile for 2-Butanone (PB93-110708,
1992) 144 pp.
Properties
Flammable liquid; acetone-like odor. d420 0.805. mp 86°. bp 79.6°. Flash
pt, closed cup: 21°F (6°C). nD15 1.3814. nD25 1.3782. Sol in ≈4 parts water (27.5%); less sol at higher temp; miscible
with alcohol, ether, benzene. Constant boiling mixture with water, bp 73.4°, contains 88.7%
methyl ethyl ketone. Soly of water in methyl ethyl ketone: 12.5%
at 25°. LD50 orally in rats: 6.86 ml/kg (Smyth).Caution
Potential symptoms of overexposure
are irritation of skin, eyes and nose; dermatitis; headache; dizziness;
vomiting. See NIOSH Pocket Guide
to Chemical Hazards (DHHS/NIOSH 97-140, 1997) p 36.Use
As organic solvent; in the surface coating industry;
manuf smokeless powder; colorless synthetic resins.
