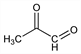
6081. Methylglyoxal
Nomenclature
Description and references
Physiological metabolite formed by the fragmentation of triosephosphates and by the metabolism of acetone; synthesized by microorganisms. Also occurs in a variety of foods and beverages such as coffee. Reacts with nucleic acids and proteins under physiological conditions serving as a signal for their degradation. Prepn: H. v. Pechmann, Ber. 20, 3213 (1887); H. O. L. Fischer, C. Taube, ibid. 57, 1502 (1924); eidem, ibid. 59, 857 (1926); M. W. Kellum et al., Anal. Biochem. 85, 586 (1978). GC determn in food and biological samples: S. Ohmori et al., J. Chromatogr. 415, 221 (1987). Review of natural occurrance and metabolism in microorganisms: Y. Inoue, A. Kimura, Adv. Microb. Physiol. 37, 177-227 (1995). Review of physiological significance and role in pathogenesis: P. J. Thornalley, Gen. Pharmacol. 27, 565-573 (1996).