6111. Methylprednisolone
Nomenclature
CAS number: 83-43-2
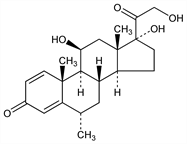
(6α,11β)-11,17,21-Trihydroxy-6-methylpregna-1,4-diene-3,20-dione; 1-dehydro-6α-methylhydrocortisone; Δ1-6α-methylhydrocortisone; 6α-methyl-11β,17α,21-triol-1,4-pregnadiene-3,20-dione; Medrate (Pharmacia & Upjohn); Medrol (Pharmacia & Upjohn); Medrone (Pharmacia). C22H30O5; mol wt 374.47.
C 70.56%, H 8.07%, O 21.36%.
Description and references
Prepn: Spero et al., J. Am. Chem. Soc. 78, 6213 (1956); Fried, ibid. 81, 1235 (1959); Sebek, Spero, US 2897218 (1959 to Upjohn); Gould, US 3053832 (1962 to Schering). Review of clinical toxicology: J. D. Truwit, Crit. Care Clin. 7, 639-657 (1991); of neuroprotective pharmacology: E. D. Hall, J. Neurosurg. 76, 13-22 (1992); of pharmacokinetics and clinical efficacy in multiple sclerosis: O. R. Hommes et al., Mult. Scler. 1, 327-328 (1996). Clinical trial in acute spinal cord injury: M. B. Bracken et al., J. Neurosurg. 89, 699 (1998); in carpel tunnel syndrome: J. W. H. H. Dammers et al., Br. Med. J. 319, 884 (1999).
Properties
Crystals, mp 228-237°. [α]D20 +83° (dioxane). uv max (95% ethanol): 243 nm (αM 14875). Sparingly sol in alcohol, dioxane, methanol; slightly sol in acetone, chloroform; very slightly sol in ether. Practically insol in water.Derivative
21-Acetate.Nomenclature
CAS number: 53-36-1
Depo-Medrol (Pharmacia & Upjohn); Depo-Medrone (Pharmacia); Vetacortyl (Vétoquinol). C24H32O6; mol wt 416.51.
C 69.21%, H 7.74%, O 23.05%.
Properties
Crystals, mp 205-208°. [α]D20 +101° (dioxane). uv max (95% ethanol): 243 nm (αM 14825). Sol in dioxane; sparingly sol in acetone, alcohol, chloroform, methanol; slightly sol in ether. Practically insol in water.Derivative
21-Succinate sodium salt.Nomenclature
CAS number: 2375-03-3
Solu-Medrol (Pharmacia & Upjohn); Solu-Medrone (Pharmacia). C26H33NaO8; mol wt 496.53.
C 62.89%, H 6.70%, Na 4.63%, O 25.78%.
Properties
White, or nearly white, odorless, hygrosopic, amorphous solid. Very sol in water, alcohol; very slightly sol in acetone. Insol in chloroform.Derivative
Aceponate.Nomenclature
CAS number: 86401-95-8
Advantan (Schering AG). C27H36O7; mol wt 472.57.
C 68.62%, H 7.68%, O 23.70%.
Therapeutic Category
Glucocorticoid.
Therapeutic Category (Veterinary)
Glucocorticoid.Keywords
Glucocorticoid