6161. Metyrosine
Nomenclature
Description and references
Inhibitor of tyrosine hydroxylase, the enzyme catalyzing the first reaction in catecholamine biosynthesis (the hydroxylation of tyrosine to dopa). Prepn: NL 6607757 (1966 to Merck & Co.), C.A. 67, 91108p (1967). Prepn of dl-form: Stein et al., J. Am. Chem. Soc. 77, 700 (1955); Potts, J. Chem. Soc. 1955, 1632; Pfister, Stein, US 2868818 (1959 to Merck & Co.); Saari, J. Org. Chem. 32, 4074 (1967). HPLC determn in serum: M. M. Hefnawy, J. T. Stewart, J. Liq. Chromatogr. Relat. Technol. 20, 3009 (1997). Chiral resolution by CE and micellar electrokinetic capillary chromatography: idem, ibid. 28, 439 (2005). Clinical pharmacology and metabolism: Engelman et al., J. Clin. Invest. 47, 568, 577 (1968). Review of pharmacology and clinical use: R. N. Brogden et al., Drugs 21, 81-89 (1981); of use prior to surgical resection of pheochromocytoma: R. R. Perry et al., Ann. Surg. 212, 621-628 (1990).
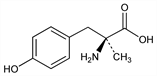
Properties
White crystals, mp 310-315°. Sol in acidic aq solns and alkaline aq soln, although subject to oxidative degradation in the latter; very slightly sol in water, acetone, and methanol. Insol in chloroform and benzene.Derivative
dl-Form.Nomenclature
Properties
Crystals from water, dec 320° (Stein); also reported as dec 330-332° (Potts). Soly in water at room temp: 0.57 mg/ml.Therapeutic Category
Antihypertensive in pheochromocytoma.
Keywords
Antipheochromocytoma