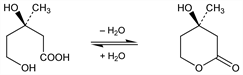6163. Mevalonic Acid
Nomenclature
Description and references
Precursor in the biosynthesis of cholesterol. Occurs in equilibrium with the δ-lactone. Isoln from distillers' solns: Wright et al., J. Am. Chem. Soc. 78, 5273 (1956). Synthesis: Wolf et al., ibid. 79, 1486 (1957); Hoffmann et al., ibid. 2316; Eggerer et al., Ann. 608, 71 (1957); US 2915398; US 2915531; US 2915532; US 2915533; US 2915551 (all 1959 to Merck & Co.); Shunk et al., US 3014963 (1961 to Merck & Co.); Hulcher, Hosick, US 3119842 (1964); F. C. Huang et al., J. Am. Chem. Soc. 97, 4144 (1975). Resolution: Shunk et al., ibid. 79, 3294 (1957); US 2945059 (1960 to Merck & Co.). Synthesis of the δ-lactone: R. H. Cornforth et al., Tetrahedron 18, 1351 (1962); E. L. Eliel, K. Soai, Tetrahedron Lett. 22, 2859 (1981); A. Banerji, G. P. Kalena, Synth. Commun. 12, 225 (1982). Review of organic and biochemistry: Wagner, Folkers, Endeavour 20, 177-187 (Oct. 1961). Prepn of 5-phosphate: Robinson, Wittreich, US 3014057 (1961 to Merck & Co.). Review of role of mevalonic acid in sterol biosynthesis: G. J. Schroepfer, Jr., Annu. Rev. Biochem. 50, 585-621 (1981); E. Caspi, Tetrahedron 42, 3-50 (1986).