6188. Miglustat
Nomenclature
Description and references
Amino sugar; inhibitor of glucosyltransferase, an enzyme involved in the biosynthesis of glycosphingolipids. Prepn: B. Junge et al., DE 2758025; see also, eidem, US 4639436 (1979, 1987 both to Bayer). Improved synthesis: E. W. Baxter, A. B. Reitz, J. Org. Chem. 59, 3175 (1994); C. R. R. Matos et al., Synthesis 1999, 571. In vitro efficacy vs HIV: A. Karpas et al., Proc. Natl. Acad. Sci. USA 85, 9229 (1988). Inhibition of glycolipid biosynthesis: F. M. Platt et al., J. Biol. Chem. 269, 8362 (1994). Clinical evaluation in Gaucher's disease: T. Cox et al., Lancet 355, 1481 (2000). Review of pharmacology and clinical development in Gaucher's disease: P. L. McCormack, K. L. Goa, Drugs 63, 2427-2434 (2003).
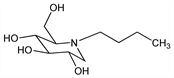
Properties
White to off-white crystalline solid, mp 125-126°. [α]D25 15.9° (c = 0.77 in water). Highly sol in water (>1000 mg/ml).Therapeutic Category
In treatment of inherited glycosphingolipid lysosomal storage disorders.