6210. Misoprostol
Nomenclature
Description and references
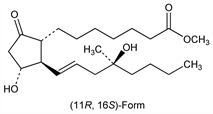
Cytoprotective prostaglandin PGE1 analog; also exhibits uterotonic and cervical-ripening actions. Double racemate comprised of the (+)- and (-)-enantiomers of the 16R- and 16S-forms. The pharmacologically active form is the (11R,16S)-enantiomer. Prepn: P. W. Collins, R. Pappo, BE 827127; eidem, US 3965143 (1975, 1976 both to Searle); P. W. Collins et al., Tetrahedron Lett. 48, 4217 (1975). Prepn, activity, NMR data: P. Collins et al., J. Med. Chem. 20, 1152 (1977). HPLC resolution of enantiomers: D. A. Roston, R. Wijayaratne, Anal. Chem. 60, 948 (1988). Mechanism of gastric secretory inhibition: D. G. Colton et al., Arch. Int. Pharmacodyn. Ther. 236, 86 (1978). Symposium on pharmacology and clinical efficacy: Dig. Dis. Sci. 30, Suppl. 11, 114S-205S (1985). Toxicology profile: F. N. Kotsonis et al., ibid. 142S. Clinical trial in prevention of NSAID-induced ulcer: D. Y. Graham et al., Ann. Intern. Med. 119, 257 (1993); to induce labor: H. Fletcher et al., Obstet. Gynecol. 83, 244 (1994). Review of clinical experience in pregnancy: A. B. Goldberg et al., N. Engl. J. Med. 344, 38-47 (2001).
Properties
Light yellow oil. Sol in water. LD50 in rats, mice (mg/kg): 40-62, 70-160 i.p.; 81-100, 27-138 orally (Kotsonis).Therapeutic Category
Antiulcerative.
Therapeutic Category (Veterinary)
Antiulcerative.Keywords
Antiulcerative; Prostaglandin/Prostaglandin Analog