6222. Mizoribine
Nomenclature
Description and references
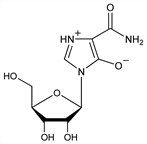
Nucleoside antibiotic produced by Eupenicillium brefedianum with cytotoxic and immunosuppressive activity. Isoln: K. Mizuno et al., BE 799805; eidem, US 3888843 (1973, 1975 to Toyo Jozo); eidem, J. Antibiot. 27, 775 (1974). Synthesis: M. Hayashi et al., Chem. Pharm. Bull. 23, 245 (1975); K. Fukukuwa et al., ibid. 32, 1644 (1984). Prepn of the aglycone: T. Atsumi et al., JP Kokai 76 88965 (1976 to Sumitomo), C.A. 86, 106582g (1977). HPLC determn in human serum: K. Takada et al., J. Chromatogr. 222, 156 (1981). Anti-arthritic activity: H. Iwata et al., Experientia 33, 502 (1977). Cytotoxic effect and comparison with aglycone: K. Sakaguchi et al., J. Antibiot. 28, 798 (1975). Antitumor spectrum of aglycone: N. Yoshida et al., Cancer Res. 43, 5851 (1983). Pharmacokinetics: K. Takada, Eur. J. Pharmacol. 24, 457 (1983). Clinical trials in renal transplantation: T. Inou et al., Transplant. Proc. 12, 526 (1980); eidem, ibid. 13, 315 (1981); A. Tajima et al., Transplantation 38, 116 (1984).
Properties
Crystals from methanol, mp >200° (dec). [α]D27 -35° (c = 0.8 in H2O). pKa 6.75. uv max (H2O): 245, 279 nm (E 250, 580). Sol in water. Slightly sol in methanol, ethanol. Insol in most organic solvents. LD50 in mice (g/kg): >1.5 i.v., >2.4 i.p. (Mizuno, 1975).Derivative
Aglycone.Nomenclature
5-Hydroxy-1H-imidazole-4-carboxamide; 4-carbamoyl-5-hydroxyimidazole; 4-carbamoylimidazolium-5-olate; SM-108.Therapeutic Category
Immunosuppressant.
Keywords
Immunosuppressant