6224. αMNP
Nomenclature
CAS number: 63628-25-1
α-Methoxy-α-methyl-1-naphthaleneacetic acid; 2-methoxy-2-(1-naphthyl)propanoic acid. C14H14O3; mol wt 230.26.
C 73.03%, H 6.13%, O 20.85%.
Description and references
Chiral derivatization reagent. Prepn and application to amino acids: J. Goto et al., Chem. Pharm. Bull. 25, 847 (1977); eidem, J. Chromatogr. 152, 413 (1978). Resolution of enantiomers and abs config: N. Harada et al., Tetrahedron: Asymmetry 11, 1249 (2000). Use in alcohol resolution: A. Ichikawa, Chirality 11, 70 (1999).
Properties
Colorless plates from acetone-hexane, mp 161-162°. uv max (methanol): 263, 271, 281, 287, 292 nm (log ε 3.36, 3.53, 3.57, 3.46, 3.45).Derivative
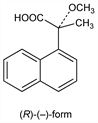
(-)-(R)-Form.Nomenclature
CAS number: 63628-26-2
Properties
Colorless plates from ether-hexane, mp 111-112°. [α]D13 -106.3° (c = 0.16 in CHCl3); [α]D13 -128.8° (c = 0.10 in methanol).Derivative
(+)-(S)-Form.Nomenclature
CAS number: 102691-93-0
Properties
[α]D26 +67.4° (c = 1.39 in CHCl3).Derivative
Hydroxy acid.Nomenclature
CAS number: 6341-58-8 (unspecified stereo);
255881-88-0
(S-(+)-form)
α-Hydroxy-α-methyl-1-naphthaleneacetic acid; αHNP. C13H12O3; mol wt 216.23.
C 72.21%, H 5.59%, O 22.20%.
Description and references
Prepn and abs config: A. Ichikawa et al., Tetrahedron: Asymmetry 10, 4075 (1999).