6229. Moexipril
Nomenclature
CAS number: 103775-10-6
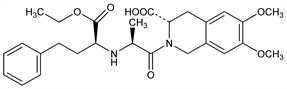
(3S)-2-[(2S)-2-[[(1S)-1-(Ethoxycarbonyl)-3-phenylpropyl]amino]-1-oxopropyl]-1,2,3,4-tetrahydro-6,7-dimethoxy-3-isoquinolinecarboxylic
acid; RS-10085. C27H34N2O7; mol wt 498.57.
C 65.04%, H 6.87%, N 5.62%, O 22.46%.
Description and references
Angiotensin converting enzyme (ACE) inhibitor; dimethoxy analog of quinapril, q.v. Prepn: M. L. Hoefle, S. Klutchko, EP 49605; eidem, US 4344949 (both 1982 to Warner-Lambert); S. Klutchko et al., J. Med. Chem. 29, 1953 (1986). Pharmacology: O. Edling et al., J. Pharmacol. Exp. Ther. 275, 854 (1995). GC-MS determn in plasma: W. Hammes et al., J. Chromatogr. B 670, 81 (1995). Clinical trials in hypertension: W. B. White et al., J. Hum. Hypertens. 8, 917 (1994); M. Stimpel et al., Cardiology 87, 313 (1996).
Derivative
Hydrochloride.Nomenclature
CAS number: 82586-52-5
CI-925; RS-10085-197; SPM-925; Fempress (Schwarz); Perdix (Schwarz); Univasc (Schwarz). C27H34N2O7.HCl; mol wt 535.03.
C 60.61%, H 6.59%, N 5.24%, O 20.93%, Cl 6.63%.
Properties
Crystals from ethanol + ethyl ether, mp 141-161°. [α]D23 +34.2° (c = 1.1 in ethanol).Derivative
Diacid hydrochloride.Nomenclature
CAS number: 82586-57-0
Moexiprilat hydrochloride. C25H30N2O7.HCl; mol wt 506.98.
C 59.23%, H 6.16%, N 5.53%, O 22.09%, Cl 6.99%.
Properties
Prepd as the monohydrate; crystals from THF + ethanol, mp 145-170°. [α]D23 +37.8° (c = 1.1 in methanol).Therapeutic Category
Antihypertensive.
Keywords
ACE-Inhibitor; Antihypertensive; N-Carboxyalkyl (peptide/lactam) Derivatives