6280. Mosher's Reagent
Nomenclature
CAS number: 81655-41-6
α-Methoxy-α-(trifluoromethyl)benzeneacetic acid; (±)-Mosher's acid; MTPA; 2-methoxy-2-(trifluoromethyl)phenylacetic acid. C10H9F3O3; mol wt 234.17.
C 51.29%, H 3.87%, F 24.34%, O 20.50%.
Description and references
Chiral derivatizing reagent. Prepn: D. L. Dull, H. S. Mosher, J. Am. Chem. Soc. 89, 4230 (1967); J. A. Dale et al., J. Org. Chem. 34, 2543 (1969). Microscale prepn: D. E. Ward, C. K. Rhee, Tetrahedron Lett. 32, 7165 (1991). Absolute configuration: S. S. Oh et al., J. Org. Chem. 54, 4499 (1989); of the acid chloride: B. S. Joshi, S. W. Pelletier, Heterocycles 51, 183 (1999). Enantiomeric purity of commerical samples: W. A. K"onig et al., Tetrahedron Lett. 31, 6867 (1990). Use in determn of abs config: T. R. Hoye, M. K. Renner, J. Org. Chem. 61, 2056 (1996); A. Ichikawa, Enantiomer 3, 255 (1998); L. F. Tietze et al., Eur. J. Org. Chem. 2000, 2247.
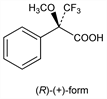
Derivative
(R)-(+)-Form.Nomenclature
CAS number: 20445-31-2
Properties
bp1.5 mm 116-118°. [α]D25 +68.5 ± 1.3° (c = 1.49 in CH3OH).Derivative
(S)-(-)-Form.Nomenclature
CAS number: 17257-71-5
Properties
bp1.5 mm 115-117°. [α]D24 -71.8± 0.6° (c = 3.28 in CH3OH).Derivative
(R)-(-)-Acid chloride.Nomenclature
CAS number: 39637-99-5
(R)-MTPA-chloride. Properties
bp1 mm 54-56°. [α]D25 -10.0± 0.1° (neat, l = 1).Derivative
(S)-(+)-Acid chloride.Nomenclature
CAS number: 20445-33-4