6291. Moxifloxacin
Nomenclature
CAS number: 151096-09-2
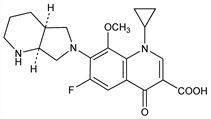
1-Cyclopropyl-6-fluoro-1,4-dihydro-8-methoxy-7-[(4aS,7aS)-octahydro-6H-pyrrolo[3,4-b]pyridin-6-yl]-4-oxo-3-quinolinecarboxylic acid. C21H24FN3O4; mol wt 401.43.
C 62.83%, H 6.03%, F 4.73%, N 10.47%, O 15.94%.
Description and references
Fluorinated quinolone antibacterial. Prepn: U. Petersen et al., EP 550903 (1993 to Bayer). Antibacterial spectrum in vitro: J. M. Woodcock et al., Antimicrob. Agents Chemother. 41, 101 (1997). Activity vs Mycobacterium tuberculosis: B. Ji et al., ibid. 42, 2066 (1998). HPLC determn in serum: C. M. Tobin et al., J. Antimicrob. Chemother. 42, 278 (1998). Clinical pharmacokinetics: H. Stass et al., Antimicrob. Agents Chemother. 42, 2060 (1998). Review of pharmacology and clinical experience: M. Miravitlles, Expert Opin. Pharmacother. 6, 283-293 (2005).
Properties
mp 203-208° (dec). [α]23D -193°.Derivative
Hydrochloride.Nomenclature
CAS number: 186826-86-8
Bay-12-8039; Actimax (Sankyo); Actira (Bayer); Avalox (Bayer); Avelox (Bayer); Octegra (Bayer); Proflox (Esteve); Vigamox (Alcon). C21H24FN3O4.HCl; mol wt 437.89.
C 57.60%, H 5.75%, F 4.34%, N 9.60%, O 14.61%, Cl 8.10%.
Properties
Slightly yellow to yellow crystalline powder, mp 324-325° (dec). [α]25D -256° (c = 0.5 in water).Therapeutic Category
Antibacterial.
Keywords
Antibacterial (Synthetic); Quinolones and Analogs