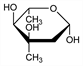
6318. Mycarose
Nomenclature
Description and references
Constituent of the macrolides carbomycin A and B, spiramycin A, B and C and tylosin: Woodward, Angew. Chem. 69, 50 (1957); Paul, Tchelitcheff, Bull. Soc. Chim. Fr. 1957, 443; 1960, 150; Hamill et al., Antibiot. Chemother. 11, 328 (1961). Part of carbomycin molecule: Regna et al., J. Am. Chem. Soc. 75, 4625 (1953). Stereochemistry: Grisebach, Hofheinz, Angew. Chem. 74, 499 (1962); Foster et al., Proc. Chem. Soc. London 1962, 254; Hofheinz et al., Tetrahedron 18, 1265 (1962). Synthesis of dl-mycarose: Korte et al., ibid. 1257; Grisebach et al., Ber. 96, 1823 (1963). Synthesis of l-mycarose: Lemal et al., Tetrahedron 18, 1275 (1962); Koto et al., Bull. Chem. Soc. Jpn. 45, 532 (1972). Synthesis of d-mycarose: Flaherty et al., J. Chem. Soc. C 1966, 398. Isoln from aureolic acid: Berlin et al., Khim. Prir. Soedin. 8, 535 (1972), C.A. 78, 4432a (1973).