6342. Nabilone
Nomenclature
Description and references
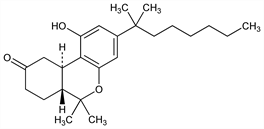
Synthetic cannabinoid with antiemetic, antiglaucoma, and CNS activity. Prepn: R. A. Archer, DE 2451934, idem, US 3968125 (1975, 1976 both to Lilly); R. A. Archer et al., J. Org. Chem. 42, 2277 (1977). Pharmacology: P. Stark, R. A. Archer, Pharmacologist 17, 210 (1975); R. M. Orzelek-O'Neil et al., Toxicol. Appl. Pharmacol. 54, 493 (1980). Physiological disposition: A. Rubin et al., Clin. Pharmacol. Ther. 22, 85 (1977). Pharmacokinetics: H. R. Sullivan et al., Biomed. Mass Spectrom. 5, 296 (1978). Behavioral effects: P. Stark, P. B. Dews, J. Pharmacol. Exp. Ther. 214, 124 (1980). Clinical studies as antiemetic in cancer patients: N. Steele et al., Cancer Treat. Rep. 64, 219 (1980); C. J. Williams et al., Cancer Clin. Trials 3, 363 (1980). Clinical evaluation of anxiolytic effects: L. F. Fabre et al., J. Clin. Pharmacol. 21, Suppl 8-9, 377S (1981); R. M. Glass et al., ibid. 383S. Review of pharmacology and efficacy as antiemetic: A. Ward, B. Holmes, Drugs 30, 127-144 (1985). Toxicity study in neonatal rats: C. L. Moss et al., Toxicol. Appl. Pharmacol. 48, A120 (1979). Comprehensive description: R. W. Souter, Anal. Profiles Drug Subs. 10, 499-512 (1981).
Properties
White crystals from ethyl acetate/hexane, mp 159-160°. uv max (ethanol): 207, 280 nm (ε 47000, 250). pKa in 66% DMF: 13.5.Note
This is a controlled substance (hallucinogen): 21 CFR, 1308.12.Therapeutic Category
Antiemetic.
Keywords
Antiemetic