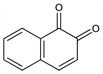
6394. 1,2-Naphthoquinone
Nomenclature
CAS number: 524-42-5
1,2-Naphthalenedione; β-naphthoquinone. C10H6O2; mol wt 158.15.
C 75.94%, H 3.82%, O 20.23%.
Description and references
Prepd by oxidation of 1-amino-2-naphthol with ferric chloride: Fieser, Org. Synth. 17, 68 (1937); by oxidation of naphthalene: Milas, US 2395638 (1946 to Research Corp.); by bacterial dissimilation of naphthalene: Murphy, Stone, Can. J. Microbiol. 1, 579 (1955); from tetrachloro-o-benzoquinone: Horner, Dürckheimer, Z. Naturforsch. 14b, 741 (1959). Structure: Hodgson, Hathaway, Trans. Faraday Soc. 41, 115 (1945). Spectroscopic properties: Oliver et al., Tetrahedron 24, 4067 (1968).
Properties
Golden yellow needles, dec 145-147°. uv max (abs alc): 250, 340, 405 nm (log ε 4.35, 3.40, 3.40). Sol in alc, benzene, ether, 5% NaOH, 5% NaHCO3, concd H2SO4 with green color. Practically insol in water.Derivative
2-Semicarbazone.Nomenclature
CAS number: 31853-38-0
Naftazone; Haemostop Injection; Mediaven (Syntex); Karbinon (Badrial). C11H9N3O2; mol wt 215.21.
C 61.39%, H 4.22%, N 19.53%, O 14.87%.
Use
As reagent for resorcinol and thalline.Therapeutic Category
2-Semicarbazone as hemostatic.
Keywords
Hemostatic