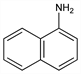
6398. 1-Naphthylamine
Nomenclature
CAS number: 134-32-7
1-Naphthalenamine; 1-aminonaphthalene; α-naphthylamine; naphthalidine. C10H9N; mol wt 143.19.
C 83.88%, H 6.34%, N 9.78%.
Description and references
Prepd by reducing α-nitronaphthalene with Fe and HCl: West, J. Chem. Soc. 127, 494 (1925); from 1-naphthalenecarboxylic acid and hydroxylamine: Snyder et al., J. Am. Chem. Soc. 75, 2014 (1953). Review of carcinogenic risk: IARC Monographs 4, 87-96 (1974).
Properties
Needles, becoming red on exposure to air, or a reddish, cryst mass; unpleasant odor. Sublimes; volatile with steam. d 1.13. mp 50°. bp 301°. Flash pt 157°C. Sol in 590 parts water; freely sol in alc, ether. Reduces warm ammoniacal silver nitrate. Keep well closed and protected from light.Derivative
Hydrochloride.C10H9N.HCl; mol wt 179.65.
C 66.86%, H 5.61%, N 7.80%, Cl 19.73%.
Properties
Cryst powder; becomes bluish on exposure to air and light. Sol in about 27 parts water; sol in alcohol, ether. Protect from light.Derivative
Sulfate dihydrate.(C10H9N)2.H2SO4.2H2O; mol wt 420.48.
C 57.13%, H 5.75%, N 6.66%, S 7.63%, O 22.83%.