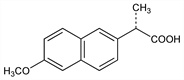
6417. Naproxen
Nomenclature
Description and references
Nonsteroidal anti-inflammatory. Prepn: J. H. Fried, I. T. Harrison, ZA 6707597; eidem, US 3904682; eidem, US 4009197 (1968, 1975, 1977 all to Syntex); I. T. Harrison et al., J. Med. Chem. 13, 203 (1970). Pharmacology: Roszkowski et al., J. Pharmacol. Exp. Ther. 179, 114 (1971). Activity may be due to the ability to inhibit prostaglandin biosynthesis. Mode of action studies: Tomlinson et al., Biochem. Biophys. Res. Commun. 46, 552 (1972). Metabolism: Runkel et al., J. Pharm. Sci. 61, 703 (1972). HPLC determn in plasma and serum: P. J. Streete, J. Chromatogr. 495, 179 (1989). Stereoselective synthesis: K. T. Wan, M. E. Davis, Nature 370, 449 (1994). Clinical studies: Katona et al., Clin. Trials J. 8, 3 (1972); Runkel, Chem. Pharm. Bull. 20, 1457 (1972). Review: Arzneim.-Forsch. 25, 278-332 (1975). Review of pharmacology and therapeutic efficacy: R. N. Brogden et al., Drugs 18, 241-277 (1979). Comprehensive description: F. J. Al-Shammary et al., Anal. Profiles Drug Subs. Excip. 21, 345-373 (1992).
Properties
Crystals from acetone-hexane, mp 152-154°. [α]D +66° (in chloroform). Sol in 25 parts ethanol (96%), 20 parts methanol, 15 parts chloroform, 40 parts ether. Practically insol in water. LD50 in mice (mg/kg): 435 i.v.; 1234 orally; in rats (mg/kg): 575 i.p.; 534 orally (Roszkowski).Derivative
Piperazine salt.Nomenclature
Derivative
Sodium salt.Nomenclature
Properties
Crystals from acetone, mp 244-246°. [α]D -11° (in methanol).Therapeutic Category
Anti-inflammatory; analgesic; antipyretic.
Therapeutic Category (Veterinary)
Anti-inflammatory.Keywords
Anti-inflammatory (Nonsteroidal); Arylpropionic Acid Derivatives; Analgesic; Non-opioids; Acetic/Propionic Acid Derivatives; Antipyretic