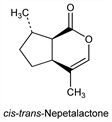
6470. Nepetalactone
Nomenclature
Description and references
The first fully characterized methylcyclopentane monoterpenoid. Isolated from the volatile oil of catnip produced by Nepeta cataria L., Labiatae: S. M. McElvain et al., J. Am. Chem. Soc. 63, 1558 (1941); J. Meinwald, ibid. 76, 4571 (1954). First believed to be a single entity, it was later shown to be a mixture of the cis-trans and trans-cis isomers, the cis-trans isomer comprising 70-99% of the oil. Structure: S. M. McElvain, E. J. Eisenbraun, J. Am. Chem. Soc. 77, 1599 (1955). Configuration of the cis-trans and trans-cis isomers: R. B. Bates et al., ibid. 80, 3420 (1958). Separation and properties: R. B. Bates, C. W. Siegel, Experientia 19, 564 (1963). Biosynthesis: F. E. Regnier et al., Phytochemistry 7, 221 (1968). Metabolism in cats: G. R. Waller et al., Science 164, 1281 (1969). Behavioral and toxicological study: J. W. Harney et al., Lloydia 41, 367 (1978). Structural study of isomeric nepetalactones: E. J. Eisenbraun et al., J. Org. Chem. 45, 3811 (1980).