6527. Nicotinyl Alcohol
Nomenclature
CAS number: 100-55-0
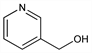
3-Pyridinemethanol; β-pyridylcarbinol; 3-hydroxymethylpyridine; nicotinic alcohol; Nu-2121; Roniacol (Roche); Ronicol. C6H7NO; mol wt 109.13.
C 66.04%, H 6.47%, N 12.83%, O 14.66%.
Description and references
Prepd by catalytic hydrogenation of 3-pyridinecarboxaldehyde: Panizzon, Helv. Chim. Acta 24, supplemental issue in honor of Gadient Engi, p 26E (1941); by lithium aluminum hydride reduction of ethyl nicotinate: Rosenmund, Zymalkowski, Ber. 85, 156 (1952); Cohen, US 2520037 (1950); Mosher, Tessieri, J. Am. Chem. Soc. 73, 4926 (1951); of methyl nicotinate: Bohlmann, Ber. 86, 1423 (1953). From 3-cyanopyridine: Chase, US 2615896 (1952 to Hoffmann-La Roche); from 3-aminomethylpyridine: Schl"apfer, US 2547048 (1951 to Hoffmann-La Roche); from thionicotinic acid S-methyl ester: Ruzicka, Prelog, US 2509171 (1950 to Ciba).
Properties
Very hygroscopic liquid. bp28 154°; bp16 144-145°; bp12 114°; bp0.1 110°. Freely sol in water, ether. Sparingly sol in petr ether.Derivative
d-Tartrate.Nomenclature
CAS number: 6164-87-0
Roniacol Tartrate (Roche); Radecol; Niltuvin (EGYT). C6H7NO.C4H6O6; mol wt 259.21.
C 46.34%, H 5.06%, N 5.40%, O 43.21%.
Properties
Crystals, sour taste, mp 147-148°. Sol in ether, freely sol in water, alcohol.Use
Solubilizer for riboflavin: US 2458430 (1949).Therapeutic Category
Vasodilator (peripheral).
Keywords
Vasodilator (Peripheral)