6547. Nimbiol
Nomenclature
CAS number: 561-95-5
(4aS-trans)-2,3,4,4a,10,10a-Hexahydro-6-hydroxy-1,1,4a,7-tetramethyl-9(1H)-phenanthrenone; 6-hydroxy-7-methyl-9-oxopodocarpane. C18H24O2; mol wt 272.38.
C 79.37%, H 8.88%, O 11.75%.
Description and references
(+)-Form found in the bark of the neem tree, Azadirachta indica A. Juss. (Melia azadirachta L.), Meliaceae: P. Sengupta et al., Chem. Ind. (London) 1958, 861. Structure: P. Sengupta et al., Tetrahedron 10, 45 (1960). Also obtained by conversion of podocarpic acid: Bible, ibid. 11, 22 (1960); Wenkert et al., J. Am. Chem. Soc. 83, 2320 (1961). Total synthesis of (±)-form: W. L. Meyer et al., J. Org. Chem. 40, 3686 (1975). Synthesis of (±)-methyl ether: Ramachandran, Dutta, J. Chem. Soc. 1960, 4766; Delobelle, Fétizon, Bull. Soc. Chim. Fr. 1961, 1900; Nasipuri, Roy, J. Indian Chem. Soc. 40, 327 (1963).
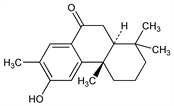
Properties
Crystals from dilute methanol or platelets by high vac sublimation. mp 248-252°. [α]D25 +33° (chloroform). uv max (abs alcohol): 234, 283 nm (log ε 4.13, 4.10).Derivative
Methyl ether.C19H26O2; mol wt 286.41.
C 79.68%, H 9.15%, O 11.17%.
Properties
Crystals, mp 143°. [α]D25 +43.7° (chloroform). uv max (alcohol): 207, 232, 279 nm (log ε 4.18, 4.15, 4.12).Derivative
(±)-Form.Properties
White needles from CH3OH, mp 237.0-237.5°.Derivative
(±)-Methyl ether.C19H26O2; mol wt 286.41.
C 79.68%, H 9.15%, O 11.17%.