6551. Nimodipine
Nomenclature
Description and references
Dihydropyridine calcium channel blocker. Prepn: H. Meyer et al., DE 2117571; eidem, US 3799934 (1972, 1974 to Bayer). Pharmacology: R. Towart, S. Kazda, Br. J. Pharmacol. 67, 409P (1979); K. Tanaka et al., Arzneim.-Forsch. 30, 1494 (1980); L. M. Auer, ibid. 31, 1423 (1981). Prepn of isomers and pharmacological comparison with racemate: R. Towart et al., ibid. 32, 338 (1982). Use as cerebral vasodilator: H. Meyer et al., GB 2018134; eidem, US 4406906 (1979, 1983 to Bayer). Effect on associative learning in aging rabbits: R. A. Deyo et al., Science 243, 809 (1989). GC and LC determns in biological fluids: G. J. Krol et al., J. Chromatogr. 305, 105 (1984). Clinical trial in prophylaxis of cerebral vasospasm: G. S. Allen et al., N. Engl. J. Med. 308, 619 (1983). Toxicology: H. Schlüter, Arzneim.-Forsch. 36, 1733 (1986). Series of articles on clinical pharmacology and therapeutic use: Am. J. Cardiol. 55(3), 139B-153B (1985).
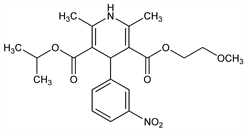
Properties
Crystals from petr ether/acetic ester, mp 125°. LD50 in mice, rats (mg/kg): 3562, 6599 orally; 33, 16 i.v. (Schlüter).Derivative
(+)-Form.Properties
[α]D20 +7.9° (c = 0.439 in dioxane).Derivative
(-)-Form.Properties
[α]D20 -7.93° (c = 0.374 in dioxane).Therapeutic Category
Vasodilator (cerebral).
Keywords
Calcium Channel Blocker; Dihydropyridine Derivatives; Vasodilator (Cerebral)