6567. Nitazoxanide
Nomenclature
Description and references
Broad spectrum antiparasitic agent; inhibits pyruvate ferredoxin oxidoreductase. Prepn: J. F. Rossignol, R. Cavier, DE 2438037; eidem, US 3950351 (1975, 1976 both to S.P.R.L. Phavic); and antiparasitic activity: R. Cavier et al., Eur. J. Med. Chem. - Chim. Ther. 13, 539 (1978). Antibacterial spectrum in vitro: L Dubreuil et al., Antimicrob. Agents Chemother. 40, 2266 (1996). Toxicology: J. R. Murphy, J.-C. Friedmann, J. Appl. Toxicol. 5, 49 (1985). Clinical pharmacokinetics: A. Stockis et al., Int. J. Clin. Pharmacol. Ther. 34, 349 (1996). Clinical trial in intestinal protozoan and helminthic infections: H. Abaza et al., Curr. Ther. Res. 59, 116 (1998). Review of mechanism of action and clinical experience: H. M. Gilles, P. S. Hoffman, Trends Parasitol. 18, 95-97 (2002); of therapeutic potential in gastrointestinal infections: A. Hemphill et al., Expert Opin. Pharmacother. 7, 953-964 (2006).
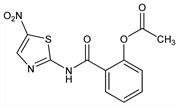
Properties
Light yellow crystalline powder. Crystals from methanol, mp 202°. Sol in DMSO (> 50mg/ml). Poorly sol in ethanol. Practically insol in water. LD50 orally in male, female mice: 1350, 1380 mg/kg; in rats: >10 g/kg (Murphy, Friedmann).Therapeutic Category
Anthelmintic (cestodes); antiprotozoal (Cryptosporidium).
Therapeutic Category (Veterinary)
In treatment of equine protozoal myeloencephalitis.Keywords
Anthelmintic (Cestodes); Antiprotozoal (Cryptosporidium)