6571. Nitracrine
Nomenclature
CAS number: 4533-39-5
N,N-Dimethyl-N′-(1-nitro-9-acridinyl)-1,3-propanediamine; 9-[[3-(dimethylamino)propyl]amino]-1-nitroacridine. C18H20N4O2; mol wt 324.38.
C 66.65%, H 6.21%, N 17.27%, O 9.86%.
Description and references
Deriv of acridine, q.v., with cytostatic and cytotoxic properties. Prepn: FR 1458183 (1966 to Polfa), C.A. 68, 39493s (1968); A. Ledochowski, B. Stefanska, Rocz. Chem. 40, 301 (1966), C.A. 65, 2219b (1966). Pharmacological studies: J. Gieldanowski et al., Arch. Immunol. Ther. Exp. 20, 399 (1972); eidem, ibid. 419. Mechanism of action: J. Konopa et al., Mater. Med. Pol. 8, 258 (1976). Cytotoxicity study: I. Szumiel, M. Walicka, Neoplasma 27, 697 (1980). DNA binding activity: L. Szmigiero, M. Gniazdowski, Arzneim.-Forsch. 31, 1875 (1981). Comprehensive review: M. Gniazdowski et al. in Antibiotics Vol. V, part 2, F. E. Hahn, Ed. (Springer-Verlag, New York, 1979) pp 275-297.
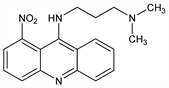
Properties
Crystals from benzene/petr ether, mp 134-135°. Practically insol in water. Sol in most organic solvents. pKa1 6.45; pKa2 8.8.Derivative
Dihydrochloride monohydrate.Nomenclature
CAS number: 55429-45-3
C-283; Ledakrin (Polfa). C18H20N4O2.2HCl.H2O; mol wt 415.31.
C 52.06%, H 5.82%, N 13.49%, O 11.56%, Cl 17.07%.
Properties
Orange crystals, mp 223-224°. Sol in water, methanol, ethanol, slightly sol in benzene, diethyl ether. Conc water solns are acidic (pH 4). LD50 in rats, mice (mg/kg): 1, 0.72 i.v.; 34, 26 i.g. (Gniazdowski).Therapeutic Category
Antineoplastic.
Keywords
Antineoplastic