C
7H
5NO
4; mol wt 167.12.
C 50.31%, H 3.02%, N 8.38%, O 38.29%.
Description and references
Prepn of m-isomer: O. Kamm, J.
B. Segur, Org. Synth. coll.
vol. I (2nd ed., 1964) p 391; of p-isomer: O. Kamm,
A. O. Matthews, ibid. p 392.
Derivative
m-Nitrobenzoic acid.
Properties
Monoclinic leaflets from water. Bitter taste.
d 1.494. mp 142°. Melts in hot water. K
(25°) = 3.48 × 10-4. Absorption spectrum: Purvis, J. Chem. Soc. 107, 971 (1915).
One gram dissolves in 320 ml water, 3 ml alc, 4 ml ether, 18 ml chloroform, ≈2
ml methanol, 2.5 ml acetone; very slightly sol in benzene, carbon
disulfide, petr ether.Derivative
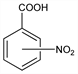
o-Nitrobenzoic acid.
Properties
Yellowish-white, intensely sweet crystals. d 1.58. mp 147-148°. One gram dissolves
in 146 ml water, 3 ml alcohol, 220 ml chloroform, 4.5 ml ether, 2.5
ml acetone, 2.5 ml methanol; very slightly sol in benzene, carbon
disulfide, petr ether.Derivative
p-Nitrobenzoic acid.
Properties
Monoclinic leaflets, plates from benzene. d 1.58. mp 242.4°. Sublimes. One gram dissolves
in 2380 ml water, 110 ml alcohol, 12 ml methanol, 150 ml chloroform,
45 ml ether, 20 ml acetone; slightly sol in benzene, carbon disulfide;
insol in petr ether.Use
Manuf intermediates; also as a reagent for alkaloids
and thorium.
