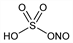6648. Nitrosylsulfuric Acid
Nomenclature
CAS number: 7782-78-7
Sulfuric acid monoanhydride with nitrous acid; nitrosyl sulfate; chamber crystals; nitrososulfuric acid; nitroxylsulfuric acid; nitrosulfonic acid; nitrosyl hydrogen sulfate; nitro acid sulfite; Nitrose (Fort Dodge). HNO5S; mol wt 127.08.
H 0.79%, N 11.02%, O 62.95%, S 25.23%.
Description and references
Formed as an intermediate in the lead chamber process for sulfuric acid by the reaction of sulfur dioxide, nitrogen trioxide, oxygen, and water: Clément, Désormes, Ann. Chim. Phys. [1] 59, 329 (1806); Lunge, J. Chem. Soc. 47, 470 (1885). Prepd from sulfur trioxide, nitrogen oxides and water: D"obereiner, Schweigger's Journ. 8, 239 (1812); de Claubry, Ann. Chim. Phys. [2] 45, 284 (1832); Kuhlmann, ibid. [3] 1, 116 (1843); from silver acid sulfate and nitrosyl bromide: Berl et al., Z. Anorg. Allg. Chem. 209, 264 (1932). See also US 1909557 and US 1909558. The formation of crystals of nitrosylsulfuric acid may be observed by igniting a mixture of 1 part sulfur and 2 or 3 parts potassium nitrate under a bell jar.