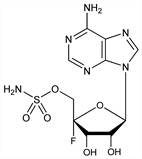
6726. Nucleocidin
Nomenclature
CAS number: 24751-69-7
4′-C-Fluoroadenosine 5′-sulfamate; 9-(4-fluoro-5-O-sulfamoylpentofuranosyl)adenine; 4′-fluoro-5′-O-sulfamoyladenosine. C10H13FN6O6S; mol wt 364.31.
C 32.97%, H 3.60%, F 5.21%, N 23.07%, O 26.35%, S 8.80%.
Description and references
Antitrypanosomal antibiotic produced by Streptomyces calvus. The first naturally occurring derivative of a fluoro sugar. Isoln and activity studies: Thomas et al., Antibiot. Annu. 1956-1957, 716; Hewitt et al., ibid. 722. Partial structure: Waller et al., J. Am. Chem. Soc. 79, 1011 (1957). Revised formula and structure: Morton et al., ibid. 91, 1535 (1969). Synthesis: Jenkins et al., ibid. 93, 4323 (1971).