6738. Obidoxime Chloride
Nomenclature
CAS number: 114-90-9
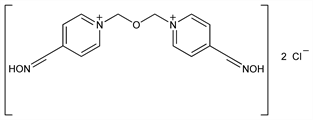
1,1′-[Oxybis(methylene)]bis[4-(hydroxyimino)methyl]pyridinium
dichloride; 1,1′-(oxydimethylene)bis[4-formylpyridinium]dichloride dioxime; N,N-dimethyleneoxidebis(pyridinium-4-aldoxime) dichloride; bis(4-hydroxyiminomethylpyridinium-1-methyl) ether dichloride; bis(isonicotinaldoxime 1-methyl) ether dichloride; BH-6; LüH6; Toksobidin (Polfa); Toxogonin (Merck KGaA). C14H16Cl2N4O3; mol wt 359.21.
C 46.81%, H 4.49%, Cl 19.74%, N 15.60%, O 13.36%.
Description and references
Cholinesterase reactivator. Prepn from pyridine aldoxime and α,α′-dichlorodimethyl ether: GB 930040; Lüttringhaus et al., US 3137702 (1963, 1964 to E. Merck); Lüttringhaus, Hagedorn, Arzneim.-Forsch. 14, 1 (1964). Pharmacology and toxicology: W. D. Erdman, H. Engelhard, ibid. 5; Mayer, Michalek, Biochem. Pharmacol. 20, 3029 (1971); Bajgar et al., Eur. J. Pharmacol. 19, 199 (1972). Hydrolysis studies: Christenson, Acta Pharm. Suec. 9, 309 (1972).
Properties
Occurs in two interchangeable isomeric forms (syn and anti). Crystals from HCl contg 70% alcohol, dec 225°. Also reported as syn, mp 235-236°; anti, mp 218-220° [Leitis et al., C.A. 71, 81098d (1969)]. Freely sol in water, stable in 1-10% aq solns. LD50 in mice, rats (mg/kg): 70, 133 i.v.; 150, 225 i.p.; >2240, >4000 orally; in mice (mg/kg): 172 i.m. (Erdman, Engelhard).Derivative
Dibromide.C14H16Br2N4O3; mol wt 448.11.
C 37.52%, H 3.60%, Br 35.66%, N 12.50%, O 10.71%.
Properties
Dec 202-203°.Therapeutic Category
Antidote (organophosphate insecticide poisoning).
Keywords
Cholinesterase Reactivator; Antidote (Organophosphate Poisoning)