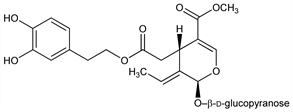
6830. Oleuropein
Nomenclature
CAS number: 32619-42-4
[2S-(2α,3E,4β)]-3-Ethylidene-2-(β-d-glucopyranosyloxy)-3,4-dihydro-5-(methoxycarbonyl)-2H-pyran-4-acetic acid 2-(3,4-dihydroxyphenyl)ethyl ester. C25H32O13; mol wt 540.51.
C 55.55%, H 5.97%, O 38.48%.
Description and references
Bitter glucoside; first secoiridoid to be isolated. Isolation from olives and the leaves and bark of the olive tree, Olea europaea L., Oleaceae and structural studies: Panizzi et al., Gazz. Chim. Ital. 90, 1449 (1960); Beyerman et al., Bull. Soc. Chim. Fr. 1961, 1821; Shasha, Leibowitz, J. Org. Chem. 26, 1948 (1961). Isoln from the ripe fruits of Ligustrum lucidum and L. japonicum Thunb, Oleaceae: Inouye, Nishioka, Tetrahedron 28, 4231 (1972). Revised structure and stereochemistry: Inouye et al., Tetrahedron Lett. 1970, 2459. Pharmacology: Petkov, Manolov, Arzneim.-Forsch. 22, 1476 (1972). Partial synthesis: A. Bianco et al., J. Nat. Prod. 55, 760 (1992).