6850. Oosporein
Nomenclature
CAS number: 475-54-7
2,2′,5,5′-Tetrahydroxy-4,4′-dimethyl[bi-1,4-cyclohexadien-1-yl]-3,3′,6,6′-tetrone; 3,3′,6,6′-tetrahydroxy-5,5′-dimethyl-2,2′-bi-p-benzoquinone; chaetomidin; iso-oosporein. C14H10O8; mol wt 306.22.
C 54.91%, H 3.29%, O 41.80%.
Description and references
Fungal pigment isolated from Oospora colorans van Beyma.: K"ogl, van Wessem, Rec. Trav. Chim. 63, 5 (1944); from Chaetonium aureum Chivers and identity with chaetomidin: Lloyd et al., J. Chem. Soc. 1955, 2163; from Acremonium spp: Divekar et al., Can. J. Chem. 37, 2097 (1959); from Beauveria bassiana (Bals.) Vuill.: Vining et al., Can. J. Microbiol. 8, 931 (1962). Identity with iso-oosporein: Smith, Thomson, Tetrahedron 10, 148 (1960). Synthesis: J. Kalamar et al., Helv. Chim. Acta 57, 2368 (1974). Biosynthesis: E. Steiner et al., ibid. 2377.
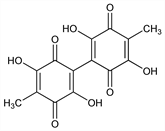
Properties
Bronze plates from aq methanol, mp 290-295°. uv max (ethanol): 216, 287 nm (log ε 3.51, 4.67).Derivative
Tetraacetate.C22H28O12; mol wt 484.45.
C 54.54%, H 5.83%, O 39.63%.
Properties
Yellow needles from methanol, mp 190°. uv max (ethanol): 262 nm (log ε 4.41).Derivative
Tetramethyl ether.C18H18O8; mol wt 362.33.
C 59.67%, H 5.01%, O 35.33%.