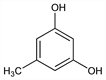
6864. Orcinol
Nomenclature
CAS number: 504-15-4
5-Methyl-1,3-benzenediol; 5-methylresorcinol; orcin; 3,5-dihydroxytoluene. C7H8O2; mol wt 124.14.
C 67.73%, H 6.50%, O 25.78%.
Description and references
Occurs in many species of lichens: Sastry, Rao, Curr. Sci. 10, 437 (1941). Prepn: Anker, Cook, J. Chem. Soc. 1945, 311; Kisteneva, Rozhdestvenskii, Zh. Prikl. Khim. 22, 1108 (1949); Stevens, US 2603662 (1952 to Gulf); Zimmer, US 3028410 (1962 to Hooker Chem.). Toxicity studies: I. Veldre et al., Vopr. Gig. Tr. Prof. Patol. Est. SSR 2, 160 (1970), C.A. 74, 51746h (1971).