6877. Orotidine
Nomenclature
CAS number: 314-50-1
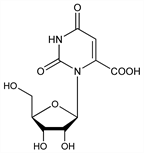
1,2,3,6-Tetrahydro-2,6-dioxo-3-β-d-ribofuranosyl-4-pyrimidinecarboxylic
acid; 3-β-d-ribofuranosylorotic acid; 6-carboxyuridine. C10H12N2O8; mol wt 288.21.
C 41.67%, H 4.20%, N 9.72%, O 44.41%.
Description and references
An orotic acid riboside obtained from cultures of Neurospora crassa mutants: Michelson et al., Proc. Natl. Acad. Sci. USA 37, 396 (1951). Isoln: Mitchell, Michelson, US 2788346 (1957 to California Inst. Res. Found.). Structure: Fox et al., Biochim. Biophys. Acta 23, 295 (1957). Synthesis: Curran, Angier, J. Org. Chem. 31, 201 (1966); US 3282919 (1966 to Am. Cyanamid). Synthesis of orotidine 5′-phosphate: Moffatt, J. Am. Chem. Soc. 85, 1118 (1963).
Properties
Needles from methanol + benzene. Turned brown near 200° but failed to melt at 400°. uv max (methanol): 268 nm (ε 8900); in 0.1N HCl: 267 nm (ε 9570); in 0.1N methanolic NaOH: 265 nm (ε 8960). Soluble in hot water, lower aliphatic alcohols and aq solns of such alcohols.Derivative
Cyclohexamine salt.C16H25N3O8; mol wt 387.39.
C 49.61%, H 6.50%, N 10.85%, O 33.04%.
Properties
Crystals from ethanol + benzene, mp 183-184°. [α]D +15° (c = 1). Also isolated as the lead salt.Derivative
Orotidine 5′-phosphate trisodium salt trihydrate.C10H10N2O11PNa3.3H2O; mol wt 488.18.
C 24.60%, H 3.30%, N 5.74%, O 45.88%, P 6.34%, Na 14.13%.
Properties
uv max (0.1N HCl): 267 nm (ε 9430). Soluble in water.Derivative
N3-Methylorotidine methyl ester.C12H16N2O8; mol wt 316.26.
C 45.57%, H 5.10%, N 8.86%, O 40.47%.