6879. Orphenadrine
Nomenclature
CAS number: 83-98-7
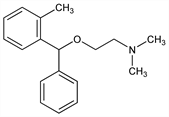
N,N-Dimethyl-2-[(2-methylphenyl)phenylmethoxy]ethanamine; N,N-dimethyl-2-(o-methyl-α-phenylbenzyloxy)ethylamine; o-methyldiphenhydramine; o-monomethyldiphenhydramine; 2-(phenyl-o-tolylmethoxy)ethyldimethylamine; phenyl-o-tolylmethyl dimethylaminoethyl ether; β-dimethylaminoethyl 2-methylbenzhydryl ether; BS-5930; Biorphen; Brocasipal. C18H23NO; mol wt 269.38.
C 80.26%, H 8.61%, N 5.20%, O 5.94%.
Description and references
Prepd by the action of 2-methylbenzhydryl chloride on dimethylaminoethanol: Bijlsma et al., Arzneim.-Forsch. 5, 72 (1955); Harms, Nauta, J. Med. Pharm. Chem. 2, 57 (1960). Covered, but not described in Rieveschl, US 2567351 (1951 to Parke, Davis). Resolution of optical isomers: van der Stelt et al., Arzneim.-Forsch. 19, 2010 (1969). Synthesis and pharmacological study of metabolites: Den Besten et al., ibid. 20, 538 (1970). Prepn of dosage forms: Harms, US 2991225 (1961 to Brocades-Stheeman).
Properties
Liquid. bp12 195°.Derivative
Hydrochloride.Nomenclature
Disipal (Doetsch, Grether); Mephenamin (Boehringer, Mann.).C18H23NO.HCl; mol wt 305.84.
C 70.69%, H 7.91%, N 4.58%, O 5.23%, Cl 11.59%.
Properties
Crystals, mp 156-157°. Sol in water, alcohol, chloroform. Sparingly sol in acetone, benzene. Practically insol in ether. pH of aq soln about 5.5.Derivative
Citrate.Nomenclature
CAS number: 4682-36-4
Banflex (Forest); Norflex (3M Pharma); X-Otag (Tutag). C18H23NO.C6H8O7; mol wt 461.50.
C 62.46%, H 6.77%, N 3.04%, O 27.73%.
Therapeutic Category
Muscle relaxant (skeletal); antihistaminic.
Keywords
Antihistaminic; Aminoalkylethers; Muscle Relaxant (Skeletal)