6901. Ouabain
Nomenclature
CAS number: 630-60-4
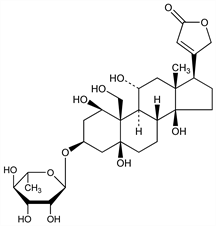
(1β,3β,5β,11α)-3-[(6-Deoxy-α-l-mannopyranosyl)oxy]-1,5,11,14,19-pentahydroxycard-20(22)-enolide; G-strophanthin; Gratus strophanthin; acocantherin. C29H44O12; mol wt 584.65.
C 59.58%, H 7.59%, O 32.84%.
Description and references
Obtained from the seeds of Strophanthus gratus (Wall. & Hock.) Baill.; also occurs in Acokanthera ouabaio Cathel and other A. spp, Apocynaceae. Isoln: Schwartze et al., J. Pharmacol. 36, 481 (1929). Hydrolysis yields one mol ouabagenin and one mol rhamnose: Jacobs, Bigelow, J. Biol. Chem. 96, 647 (1932). Toxicity study: Small et al., Toxicol. Appl. Pharmacol. 20, 44 (1971). See also Fieser, Fieser, Steroids (1959, Reinhold, New York; Chapman & Hall, London) pp 768, 772; Reichstein, Reich, Annu. Rev. Biochem. 15, 155 (1946).
Derivative
Octahydrate.Nomenclature
Purostrophan (Kali-Chemie); Strodival (Herbert); Strophoperm (Sertürner).Properties
Shiny plates (from water) which give up their water of crystn at 130°. When anhydr dec about 190°. [α]D25 -31 to -32.5° (c = 1 calcd as anhydr). Stable in air, but affected by light. One gram dissolves in about 75 ml water, in 5 ml boiling water, in 100 ml alcohol, in 8 ml boiling alcohol. Also sol in amyl alcohol, dioxane. Slightly sol in ether, chloroform, ethyl acetate. Aq solns are neutral to litmus. LD50 i.v. in rats: 14 mg/kg (Small).Therapeutic Category
Cardiotonic.
Therapeutic Category (Veterinary)
Cardiotonic, diuretic.Keywords
Cardiotonic