6955. Oxybutynin
Nomenclature
CAS number: 5633-20-5
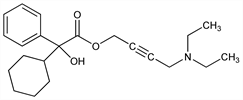
α-Cyclohexyl-α-hydroxybenzeneacetic acid 4-(diethylamino)-2-butyn-1-yl
ester; α-phenylcyclohexaneglycolic acid 4-(diethylamino)-2-butynyl
ester; 4-diethylamino-2-butynyl phenylcyclohexylglycolate; oxibutinina. C22H31NO3; mol wt 357.49.
C 73.91%, H 8.74%, N 3.92%, O 13.43%.
Description and references
Muscarinic receptor antagonist. Prepn: GB 940540 (1963 to Mead Johnson). Physico-chemical properties: E. Miyamoto et al., Analyst 119, 1489 (1994). Synthesis of S-form: S. Masumoto et al., Tetrahedron Lett. 43, 8647 (2002). GC-MS determn in plasma: K. S. Patrick et al., J. Chromatogr. 487, 91 (1989). Clinical pharmacokinetics and pharmacodynamics: J. L. Reiz et al., J. Clin. Pharmacol. 47, 351 (2007). Clinical trial in overactive bladder: P. Sand et al., BJU Int. 99, 836 (2006). Review of pharmacodynamics and therapeutic use: Y. E. Yarker et al., Drugs Aging 6, 243-262 (1995).
Properties
pKa 8.04. Log P (n-octanol/water): 2.9 (pH 6). Soly in water (mg/ml): 77 (pH 1); 0.8 (pH 6); 0.012 (pH >9.6).Derivative
Hydrochloride.Nomenclature
CAS number: 1508-65-2
Oxybutynin chloride; MJ-4309-1; Cystrin (Sanofi-Aventis); Ditropan (Sanofi-Aventis); Dridase (Sanofi-Aventis); Driptane (Solvay); Kentera (UCB); Pollakisu (Kodama); Tropax (BMS). C22H31NO3.HCl; mol wt 393.95.
C 67.07%, H 8.19%, N 3.56%, O 12.18%, Cl 9.00%.
Properties
Crystals, mp 129-130°. Sol in water, acids. Practically insol in alkali. LD50 orally in rats: 1220 mg/kg. See: E. I. Goldenthal, Toxicol. Appl. Pharmacol. 18, 185 (1971).Therapeutic Category
In treatment of urinary incontinence.
Therapeutic Category (Veterinary)
In treatment of urinary incontinence.Keywords
Antimuscarinic