6959. Oxycodone
Nomenclature
Description and references
Semisynthetic opioid analgesic. Prepn from thebaine: M. Freund, E. Speyer, J. Prakt. Chem. 94, 135 (1916); R. Krassnig et al., Arch. Pharm. 329, 325 (1996); from codeine: A. J. Walker, N. C. Bruce, Tetrahedron 60, 561 (2004). Bibliography: Small, Lutz, “Chemistry of the Opium Alkaloids,” Suppl. No. 103 to Public Health Reports, Washington (1932); K. W. Bentley, The Chemistry of the Morphine Alkaloids (Oxford, 1954). In vitro liposolubility and protein binding: R. P"oyhi"a, T. Sepp"al"a, Pharmacol. Toxicol. 74, 23 (1994). HPLC determn in serum: A. W. E. Wright et al., J. Chromatogr. B 712, 169 (1998). Review of pharmacokinetics and clinical experience: E. Kalso, J. Pain Symptom Manage. 29, S47-S56 (2005); of abuse potential: T. J. Cicero et al., J. Pain 6, 662-672 (2005).
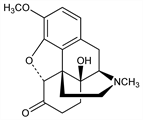
Properties
Long rods from alc, mp 218-220°. pKa 8.53. Sol in alcohol, chloroform. Nearly insol in ether, water, KOH, NaOH, NH4OH.Derivative
Hydrochloride.Nomenclature
Properties
Component of Percocet and Percodan. Long rods from water, dec 270-272°. [α]D20 -125° (c = 2.5). Partition coefficient (octanol/water): 0.7. One gram dissolves in 6-7 ml water. Slightly sol in alcohol.Derivative
Pectinate.Nomenclature
Properties
Used for prolonged action.Note
This is a controlled substance (opiate): 21 CFR, 1308.12.Therapeutic Category
Analgesic.
Keywords
Analgesic; Opioids; Phenanthrenes