6962. Oxyfedrine
Nomenclature
Description and references
Partial β-adrenergic agonist with coronary vasodilating and positive inotropic effects. Prepn: K. Thiele, BE 630296; idem, US 3225095 (1963, 1965 both to Degussa). K. Thiele et al., Arzneim.-Forsch. 16, 1064 (1966). Absolute configuration determined by circular dichroism: J. Engel et al., Chem. Ztg. 105, 85 (1981). TLC determn: Musumarra, J. Chromatogr. 350, 151 (1985). Pharmacology: H. Hueller et al., Pharmazie 27, 242 (1972). Mode of action: P. Mentz, W. Forster, Arzneim.-Forsch. 34, 1739 (1984); N. Sternitzke, Z. Kardiol. 73, 586 (1984). In prevention of experimental myocardial necrosis in rats: S. D. Seth et al., Arzneim.-Forsch. 34, 678 (1984). Comparison with atenolol, q.v., in angina pectoris: L. Fananapazir, C. Bray, Br. J. Clin. Pharmacol. 20, 405 (1985).
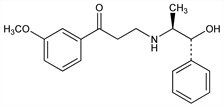
Derivative
l-Form hydrochloride.Nomenclature
Properties
Crystals from methanol, mp 192-194°. LD50 in mice (mg/kg): 29 i.v. (Hueller).Derivative
dl-Form hydrochloride.Nomenclature
Properties
mp 173-175°. LD50 in mice (mg/kg): 34 i.v. (Hueller).Therapeutic Category
Antianginal. Treatment of coronary insufficiency.
Keywords
β-Adrenergic Agonist; Antianginal; Cardiotonic