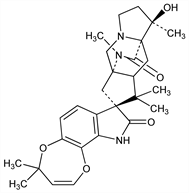
7026. Paraherquamide
Nomenclature
Description and references
Oxindole alkaloid fungal metabolite. Isolation from Penicillium paraherquei and structure: M. Yamazaki et al., Maikotokishin 10, 27 (1980), C.A. 95, 19321p (1981); M. Yamazaki et al., Tetrahedron Lett. 22, 135 (1981); from P. charlesii and nematocidal activity: J. G. Ondeyka et al., J. Antibiot. 43, 1375 (1990). Absolute stereochemistry: T. A. Blizzard et al., J. Org. Chem. 54, 2657 (1989). Approach to synthesis: R. M. Williams, T. D. Cushing, Tetrahedron Lett. 31, 6325 (1990). Anthelmintic activity in sheep: W. L. Shoop et al., J. Parasitol. 76, 349 (1990); in dogs: W. L. Shoop et al., Vet. Parasitol. 40, 339 (1991). Toxicity: eidem, Am. J. Vet. Res. 53, 2032 (1992). Mode of action: J. M. Schaeffer et al., Biochem. Pharmacol. 43, 679 (1992).