7062. Pecilocin
Nomenclature
CAS number: 19504-77-9
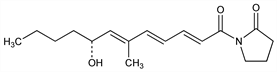
1-[(2E,4E,6E,8R)-8-Hydroxy-6-methyl-1-oxo-2,4,6-dodecatrienyl]-2-pyrrolidinone; Supral (Basotherm); Variotin (Leo Pharm). C17H25NO3; mol wt 291.39.
C 70.07%, H 8.65%, N 4.81%, O 16.47%.
Description and references
Antifungal antibiotic isolated from Paecilomyces varioti Bainier var. antibioticus: S. Takeuchi et al., J. Antibiot. 12A, 109, 195 (1959); Sumiki et al., GB 866425 (1961 to Japan. Antibiot. Res. Assoc. and Nippon Kayaku). Structure: S. Takeuchi et al., J. Antibiot. 17A, 267 (1964). Stereochemistry: S. Takeuchi, H. Yonehara, Tetrahedron Lett. 1966, 5197. Revised structure and stereochemistry (E,Z,E to E,E,E): eidem, J. Antibiot. 22, 179 (1969). Synthesis of the dl-form: A. Ishida, T. Mukaiyama, Chem. Lett. 1977, 467; eidem, Bull. Chem. Soc. Jpn. 51, 2077 (1978).
Properties
Neutral oil with ester-like odor. Does not show definite boiling or dec pt. [α]D28 -5.68° (methanol). Freely sol in methanol, ethanol, acetone, ethyl acetate, benzene, ether, chloroform, pyridine, dioxane, acetic acid; slightly sol in water, petr ether, ligroin. uv max (methanol): ca. 318, 324 nm (E1%1cm 1198). Unstable and gradually loses antifungal activity in desiccator, though it is fairly stable in organic solvents. Unstable under alkaline conditions.Derivative
Monohydrate.Properties
Needles from ethyl acetate + petr ether, mp 41.5-42.5°. uv max: 320 nm (ε 46,000).Therapeutic Category
Antifungal.
Keywords
Antifungal (Antibiotics); Polyenes