7064. Pectolinarigenin
Nomenclature
CAS number: 520-12-7
5,7-Dihydroxy-6-methoxy-2-(4-methoxyphenyl)-4H-1-benzopyran-4-one; 5,7-dihydroxy-4′,6-dimethoxyflavone; 6-methoxyacacetin; 6-hydroxypelargidenon-6,4′-dimethyl ether 1467. C17H14O6; mol wt 314.29.
C 64.97%, H 4.49%, O 30.54%.
Description and references
From leaves of Linaria vulgaris Mill., Scrophulariaceae: Schmid, Rumpel, Monatsh. Chem. 57, 421 (1931); Merz, Wu, Arch. Pharm. 274, 126 (1936). Structure: Schmid, Rumpel, Monatsh. Chem. 60, 8 (1932). Synthesis: Wessely, Moser, ibid. 56, 97 (1930); Zemplén, Farkas, Ber. 76, 937 (1943); Murti, Seshadri, Proc. Indian Acad. Sci. 30A, 78 (1949), C.A. 44, 3987d (1950); Farkas, Strelisky, Tetrahedron Lett. 1970, 187.
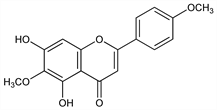
Properties
Yellow needles from methanol, mp 220-223°. uv max (methanol): 275, 335 nm. Sol in alcohol, acetone, ether, ethyl acetate. Practically insol in water, benzene, chloroform, petr ether.Derivative
Diacetate.C21H18O8; mol wt 398.36.
C 63.32%, H 4.55%, O 32.13%.
Properties
Needles from 96% alcohol, mp 151°.Derivative
7-Rutinoside.Nomenclature
Pectolinarin; neolinarin.C29H34O15; mol wt 622.57.
C 55.95%, H 5.50%, O 38.55%.