7066. Pefloxacin
Nomenclature
Description and references
Fluorinated quinolone antibacterial; analog of norfloxacin, q.v. Prepn: M. Pesson, DE 2840910; idem, US 4292317 (1979, 1981 to Roger Bellon/Dainippon). Pharmacology and antibacterial spectrum: Y. Goueffon et al., C. R. Seances Acad. Sci. Ser. 3 292, 37 (1981). Pharmacokinetics: J. Barre et al., J. Pharm. Sci. 73, 1379 (1984). Bioavailability and metabolism: A. Contrepois et al., J. Antimicrob. Chemother. 14, 51 (1984); G. Montay et al., Antimicrob. Agents Chemother. 25, 463 (1984). HPLC determn in urine and plasma: eidem, J. Chromatogr. 272, 359 (1983). Adsorptive stripping voltammetry determn in bulk form, tablets and serum: A. M. Beltagi, J. Pharm. Biomed. Anal. 31, 1079 (2003). Symposium on pharmacokinetics, clinical efficacy and safety: J. Antimicrob. Chemother. 26, Suppl. B, 1-229 (1990).
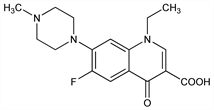
Properties
Crystals from DMF, mp 270-272° (dec). Slightly sol in water; sol in alkaline and acidic solutions. LD50 in mice (mg/kg): 225 i.v., 1000 orally; in rats (g/kg): 1.5 i.p., 2.5 orally (Goueffon).Derivative
Methanesulfonate dihydrate.Nomenclature
Therapeutic Category
Antibacterial.
Keywords
Antibacterial (Synthetic); Quinolones and Analogs